Iradimed Corp. (IRMD) is the only company in the world focused on producing IV Pumps and Patient Monitoring Systems designed for use around MRI (Magnetic Resonance Imaging) systems. Standard IV Pumps and Patient Monitors have many metal parts that cause issues around MRI systems. This necessitates disconnecting IV Pumps and Patient Monitoring and leaving them outside the MRI room or using long chords and extended IV drip lines to continue care while the patient undergoes an MRI.
This process causes a significant delay in the time it takes to prepare the patient for an MRI and can cause significant issues if mistakes are made. MRI’s are expensive to maintain and any downtime is very costly in terms of the inefficient use of these multi-million-dollar machines as well as the shell needed to operate the machine while monitoring the patient.
Company Background
The president of Iradimed, Roger Susi, has focused on solving this problem for the entirety of his career. His first company (Invivo Research) developed the first MRI compatible patient monitor in 1987. Susi sold Invivo to Intermagnetics in 2004, a large producer of standard patient monitors, which was later acquired by Philips in 2006. Philips still sells these dated monitors today, while Iradimed has drastically improved their functionality with newer generations.
Susi founded Iradimed later in 2004 with a focus on making an MRI compatible IV Pump. The company operates in a highly specialized and proprietary segment of the medical device industry, where new entrants face steep R&D requirements and costly FDA approval processes that can take years. IRMD benefits from a near-monopoly position, allowing it to command premium pricing and consistently generate gross margins above 75%.
IRMD’s original MRI-compatible IV pump, introduced in 2012, generated $26.1 million in revenue in 2024. In 2025, the company received FDA approval for its third-generation IV pump system—a major upgrade over the second generation. The new model is significantly lighter, closely resembles a standard IV pump in appearance and usability and requires minimal specialized training. It is suitable for use in all MRI procedures, with shipments expected to begin in the fourth quarter of 2025.

In 2016, IRMD launched its second generation of non-magnetic, MRI-compatible patient monitors. These monitors generated $24.4 million in revenue in 2024.
Additionally, the company earned $22.3 million in 2024 from other related products, primarily from their metal detection systems installed at MRI screening room entrances.
Quality, Proprietary Growth
With over 50,000 MRI systems installed globally and global MRI scans ranging well over 100 million annually, IRMD’s ability to provide proper monitoring and IV equipment that allow for seamless movement of patients in and out of MRI screening rooms creates a vast improvement in the amount of time it takes to conduct a screening.
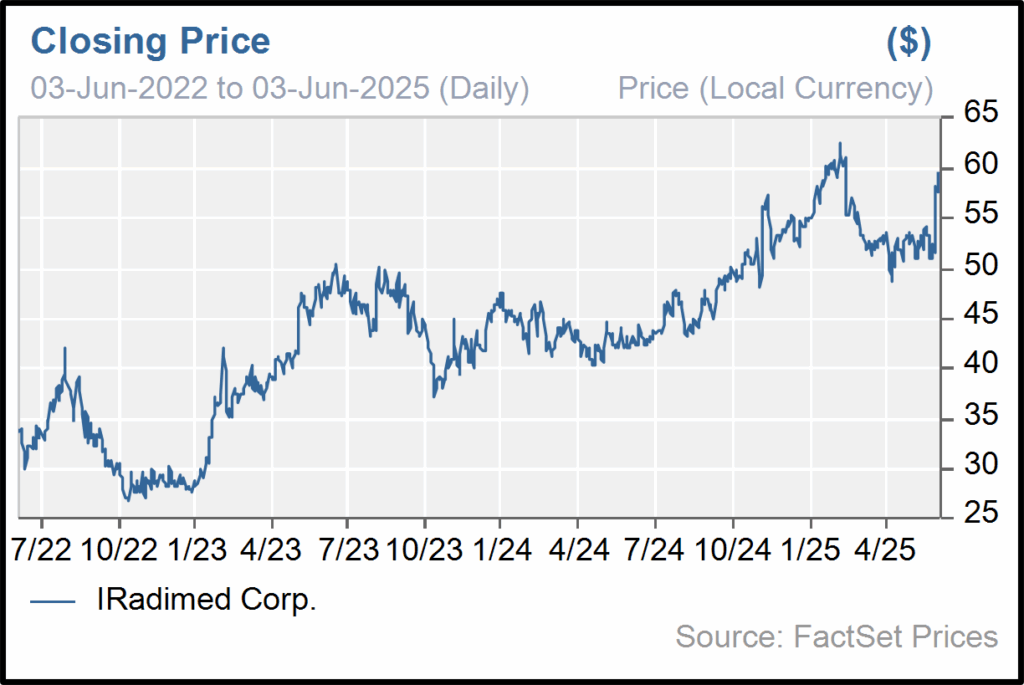
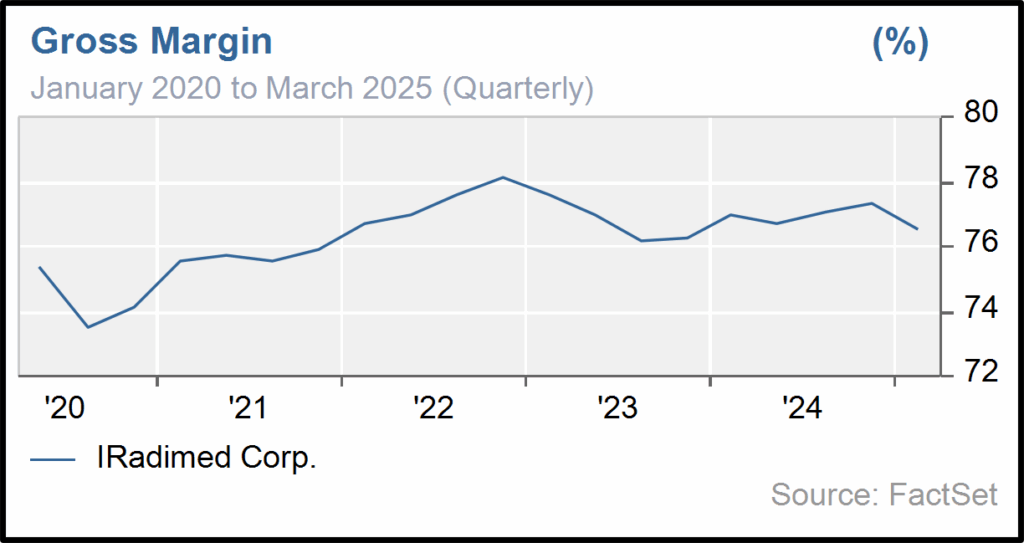
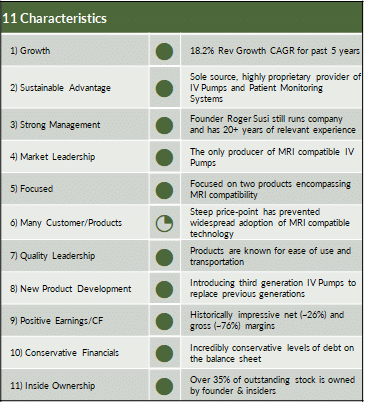
Iradimed: A Perfect Fit for Shaker
Iradimed aligns exceptionally well with Shaker’s investment philosophy, demonstrating sustained revenue growth (18.2% CAGR over 5 years), strong gross margins consistently above 70%, and quarterly EPS growth driven by pricing power and operational leverage. We expect revenue growth to accelerate as new generation of pumps replace first generation pumps at the end of their useful lives. Further, we believe there is a real opportunity as hospitals with IRMD pumps will buy more given the ease-of-use of the newer generation and IRMD will continue to penetrate hospitals and enter new markets. With no competitors in the MRI-compatible IV pump market and a patented product line, IRMD enjoys a defensible market position and clear path to continued dominance in a specialized, essential segment of hospital care. The company generates recurring, stable revenue from a growing installed base, while maintaining positive earnings, strong cash flow, and conservative financials with nearly no long-term debt. IRMD also boasts over 30% insider ownership and continues to innovate around its core technologies, supporting long-term organic growth and potential for strategic acquisition.
Risks
- IRMD competes with Philips in their MRI compatible patient screening systems segment.
- IRMD’s IV pumps sell at a significant premium to traditional IV pumps, potentially reducing demand from smaller hospitals.
- IRMD ‘s revenue is reliant on the market for MRI compatible patient monitors and IV pumps. If any disruptions occur throughout the supply chain, it may meaningfully impact future revenue streams.
This document is confidential and for the sole use of the intended original recipient. The information herein has been obtained from various public sources and we do not guarantee its accuracy. None of the information, recommendations or opinions expressed herein constitutes an offer to sell or a solicitation of an offer to buy any interest in any fund, product or security. The specific security identified and described in this report does not represent all of the securities purchased, sold or recommended for clients. It should not be assumed that investments in the securities identified and discussed will be profitable in the future. Holdings in any strategy are subject to change and actual holdings may vary by client. A list of the stocks selected for any of our strategies during the trailing twelve months is available upon request.









